Our understanding of animal behaviour depends on observation. Researchers can study how animals are born, grow and develop. We can gather evidence of how they interact with each other and their environment.
But how do we do this for extinct animals? In a recent scientific paper,
palaeontologist Zixiao Yang and colleagues compared the growth of small and giant pterosaurs.
These were flying reptiles that were alive between about 228 million years ago and 66 million years ago – sharing the Earth with dinosaurs. Yang and colleagues wanted to understand what, if anything, was different about how the giant animals got so big.

Jason Gilchrist
They looked at the limb bones, which are critical to locomotion: the forelimbs to flight, the hindlimbs to movement on the ground. With the smaller-bodied pterosaurs (the smallest in the study had a wingspan of 0.19–0.74 metres), they discovered that the limb bones that lay closest to the body – the “proximal” ones – grew more slowly relative to their total body size as the animals aged after hatching.
For large-bodied pterosaur species, such as Pteranodon, with a wingspan range of 3.91-6.37 metres, the limb bones that lay closest to the body grew faster than other elements of their skeleton after hatching.
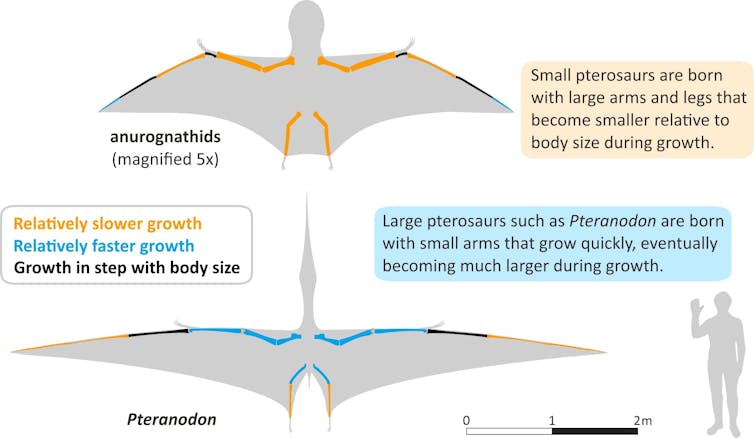
Yang Z, Jiang B, Benton MJ, Xu X, McNamara ME, Hone DWE.
In bird and mammal species alive today, this pattern is associated with particular developmental strategies. Present-day species showing a developmental pattern most similar to the smaller pterosaurs tend to move around independently from an early age.
While not necessarily lacking in parental care, such species tend to be less dependent on or demanding of their parents. By contrast, living species showing the developmental pattern seen in the larger Pteranodon tend to have young that are not capable of independent movement. In these animals, intensive parental care – including feeding the young – is the norm.
Read more:
Dinosaur egg bonanza gives vital clues about prehistoric parenting
Wing development
Using data from fossils, Yang and colleagues used computers to model the body measurements of different pterosaur species as they grew.
Pteranodon‘s wing aspect ratio (the wing length relative to wing area) increased as the species grew, allowing it to develop a long, narrow wing, associated with soaring in modern birds. The smaller pterosaurs, however, showed a consistent or decreasing wing aspect ratio during growth, allowing more manoeuvrability.
These developmental differences between larger and smaller species of pterosaur indicate that Pteranodon’s relatively greater proximal limb growth shortly after hatching, along with – perhaps – enhanced parental care, may have helped it reach a large adult size. Pterosaurs as a group encompassed the largest flying animals of all time. Hatzegopteryx thambema may have been the biggest, with a wingspan of up to 12 metres. But all pterosaurs started small.
Pterosaur hatchlings’ size was limited by ultimately the size of their eggs, which was constrained by the size of the pelvic opening of female pterosaurs, and by the soft eggshell produced by pterosaurs. Compared to hard-shelled bird eggs, soft eggs are weaker and cannot support larger sizes. To grow big, pterosaurs had to do most of their growing after they hatched.
Parental care
A key difference between the small and large species may have been parental care. This may have released large pterosaurs from growth and size constraints. An extended maturation period where parents protected their young and fed them may have allowed a bending of developmental physics, resulting in a larger body size, a lighter skeleton and more robust joints. In contrast, small pterosaur species by the nature of their slower proximal limb growth may have been locked into maturity at smaller sizes.
It’s also possible that baby pterosaurs from larger species with parental care were not capable of flight, whereas smaller species were flight-ready upon hatching.
To grow to such a large body size, the giant pterosaurs also needed two things from their environment: space and updrafts. Big pterosaurs would principally have been soarers, meaning that they used updrafts to stay aloft and economised on energy by minimising flapping. Giant pterosaurs also needed a food supply to support their large size and fuel their metabolic requirements.
While competitors for food were likely in short supply for large adult pterosaurs, youngsters – being smaller – would be more likely to overlap in terms of food sources and habitats with smaller pterosaur species. Young giant pterosaurs probably did not compete for food with adult pterosaurs of the same species.
Predation on adult giant pterosaurs by other animals would have been limited. What dinosaurs (or other creatures) would have been big and hard enough to take on such an imposing sharp-beaked monster?

Mark Witton, Author provided
Future fossils
Incredible as it seems, we can infer the presence of parental care – and lack thereof – in long-dead species of flying reptile. The odds of a pterosaur being preserved in the act of unambiguous parental care seem incredibly slim. So evidence from fossils and understanding patterns from contemporary species are critical to our understanding.
At some point, someone will hopefully find juvenile giant pterosaurs, and their hatchlings, eggs and embryos. Otherwise, questions will remain regarding the development of baby pterosaurs.
These questions include: what was the nature of the parental care? Did parents keep eggs and young warm by sitting over them? Did they defend juveniles against predators, providing food for pterosaur babies? Did males and females share parental care equally? Did they care for young that were not their own?
To more fully re-imagine the early lives and parental behaviour of giant pterosaurs, we need more fossils. Let’s find them.
![]()
Jason Gilchrist does not work for, consult, own shares in or receive funding from any company or organisation that would benefit from this article, and has disclosed no relevant affiliations beyond their academic appointment.











